
What is TESOFENSINE?
Tesofensine (NS2330) is classified as a presynaptic serotonin–noradrenaline–dopamine reuptake inhibitor. Serotonin, noradrenaline, and dopamine are all very important neurotransmitters. When these three neurotransmitters are prevented from being reabsorbed by the brain, the body responds by feeling less hungry. Tesofensine acts as an appetite suppressant while also having effects on fat oxidation and resting energy expenditure. Tesofensine also has a modulating action on dopamine which appears to affect the pleasure sensation of eating food.
Tesofensine was originally developed for the treatment of Alzheimer's and Parkinson's. However, initial studies of tesofensine for these neurological conditions wasn't successful. What researchers did find was that weight loss was consistently reported as a side effect in overweight or obese patients. Research quickly shifted and tesofensine began to be studied for weight loss.
Why is Dopamine so important?
Since tesofensine has an effect on dopamine, let's first take a look at why that matters by talking about dopamine adaptation.
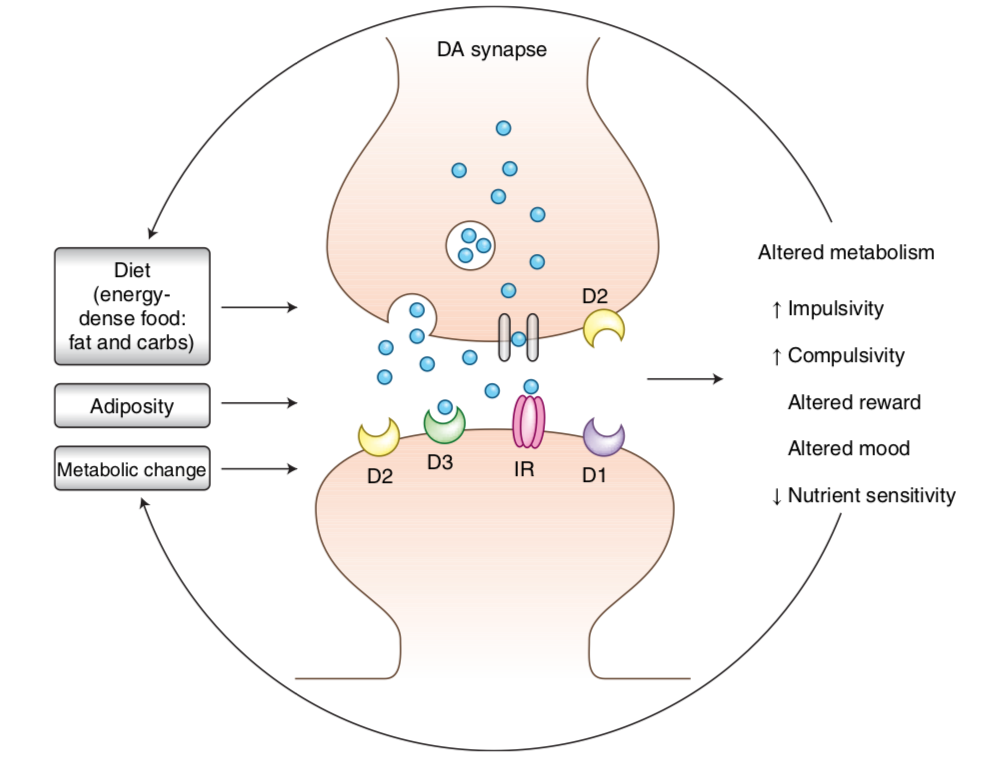
Dopamine is a neurotransmitter that helps produce feelings of satisfaction and pleasure. When it is out of balance we can lose these important benefits. Decreased dopamine signaling through overeating can lead to dopamine adaptation and sometimes behavioral changes.
This change in dopamine signaling can cause weight gain, obesity and changes in blood sugar. It is estimated that 70% of obese individuals have fewer receptors for dopamine compared to normal weight individuals. There are two reasons to explain this. Either the obese individuals have low numbers of dopamine receptors to begin with or their bodies are trying to compensate for having chronically high dopamine levels, which are triggered by chronic overeating.
How does Tesofensine affect Dopamine?
Regulation of body weight is extremely complex—it involves many physiological mechanisms and neurotransmitter action. The key is to address the dopamine receptor deficiency and find other ways to correct dopamine in obese people This can help reduce the tendency to overeat.
Dopamine transporter proteins are inhibited by tesofensine, so the dopamine receptors are stimulated for a longer period after activation and the brain's reward system is amplified. Tesofensine also increases levels of serotonin and noradrenaline. These two neurotransmitters also affect appetite and metabolism at different locations in the brain. Dopamine modulates the reward and the "pleasure" feeling of food. The other two act in the hypothalamus to increase metabolism and reduce appetite.
In addition to diet and lifestyle changes, tesofensine can help optimize dopamine response in obese patients. This combined with exercise (which we know boosts dopamine) may be a viable option for weight loss.
Tesofensine Phase 2 Study Results
The clinical Phase 2b trial reported in The Lancet showed levels of weight loss over a six-month period. Patients lost an average of 28.2lbs on a 1 mg dose, 24.9 on a 0.5 mg dose and 14.7lbs on a 0.25 mg dose compared with a 4.8lbs loss in the placebo group. All participants were instructed to follow a diet with a 300 kcal deficit and to increase their physical activity gradually to 30-60 minutes of exercise per day.
Patients being treated with tesofensine also displayed the following positive results:
- Improved levels of insulin sensitivity, higher metabolism of glucose, potentially suitable for patients at risk from type II diabetes.
- Reduction in appetite sensations within a 14-day period, greater level of satiety and less desire to eat.
- After 14 days, a significant increase in energy expenditure at rest of 6% was observed. This increase was directly linked to tesofensine and any external factors were excluded as possible causes.
- Significant increase in fat oxidation and reduction in fat tissue. The 24-hour fat oxidation increased by 15% and there was a reduction in protein oxidation.
Phase 3 study
This study was a 24-week double-blinded, randomized, placebo-controlled trial. It was designed to investigate the efficacy and safety of 0.25mg and 0.50 mg oral tesofensine compared to placebo in 372 obese patients.
What are benefits of Tesofensine?
The following are possible outcomes and benefits for individuals using tesofensine:
- Decrease in body weight and reduction in BMI
- Improvements in waist and hip circumference
- Decreased body fat
- Decreased visceral fat
- Decreased very-low-density lipoprotein & triglycerides
- Decreased insulin
- Better regulation of glucose metabolism
- Potential of reversing early type 2 diabetes
In general, tesofensine is very well tolerated with few adverse effects and very similar to placebo. When measuring cardiovascular effects, a low but statically significant increase in heart rate was noted but no significant effect on blood pressure at any of the doses tested.
The combined clinical safety database from more than 20 clinical trials with tesofensine now contains approximately 1,600 patients taking a therapeutic dose for up to a year, providing robust safety data. It has been given FDA approval as an orphan drug.
What are the Potential Side Effects of Tesofensine?
The side effects of tesofensine are similar to other currently approved diet pills and weight loss medications.
In recent trials, side effects such as dry mouth, insomnia, tachycardia, constipation, nausea, diarrhea, high blood pressure and increases in heart rate were recorded.
Get in touch if you'd like to learn more about medical weight loss.
Research
DiFeliceantonio AG, Coppin G, Rigoux L, Edwin Thanarajah S, Dagher A, Tittgemeyer M, Small DM. Supra-Additive Effects of Combining Fat and Carbohydrate on Food Reward. Cell Metab. 2018 Jul 3;28(1):33-44.e3. doi: 10.1016/j.cmet.2018.05.018. Epub 2018 Jun 14. PMID: 29909968.
Tellez LA, Medina S, Han W, Ferreira JG, Licona-Limón P, Ren X, Lam TT, Schwartz GJ, de Araujo IE. A gut lipid messenger links excess dietary fat to dopamine deficiency. Science. 2013 Aug 16;341(6147):800-2. doi: 10.1126/science.1239275. PMID: 23950538.
Koch L. Lipids: Gut-derived lipid signal links dopamine deficiency and high-fat feeding. Nat Rev Endocrinol. 2013 Nov;9(11):627. doi: 10.1038/nrendo.2013.172. Epub 2013 Aug 27. PMID: 23979314.
Bello NT, Zahner MR. Tesofensine, a monoamine reuptake inhibitor for the treatment of obesity. Curr Opin Investig Drugs. 2009 Oct;10(10):1105-16. PMID: 19777399.
Astrup A, Madsbad S, Breum L, Jensen TJ, Kroustrup JP, Larsen TM. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial. Lancet. 2008 Nov 29;372(9653):1906-1913. doi: 10.1016/S0140-6736(08)61525-1. Epub 2008 Oct 22. PMID: 18950853.
Astrup A, Meier DH, Mikkelsen BO, Villumsen JS, Larsen TM. Weight loss produced by tesofensine in patients with Parkinson's or Alzheimer's disease. Obesity (Silver Spring). 2008 Jun;16(6):1363-9. doi: 10.1038/oby.2008.56. Epub 2008 Mar 20. PMID: 18356831.
Tsai AG. Tesofensine and weight loss. Lancet. 2009 Feb 28;373(9665):719; author reply 720. doi: 10.1016/S0140-6736(09)60432-3. PMID: 19249625.
Schedule Your Consultation
Consultations are for anyone looking into their medical weight loss and appetite suppressant options. In the consultations, we work together to develop a medical weight loss plan that is safe and effective for you. Our licensed physicians will take numerous factors into consideration when finding the right option for you.
Transformyou will help you stay on track with your medical weight loss goals, checking in on you frequently regarding your general health, weight, etc. and adjusting your medical weight loss plan according to your needs. Schedule your consultation today.
All patients always work directly with one of our physicians to ensure patient safety and confidentiality.